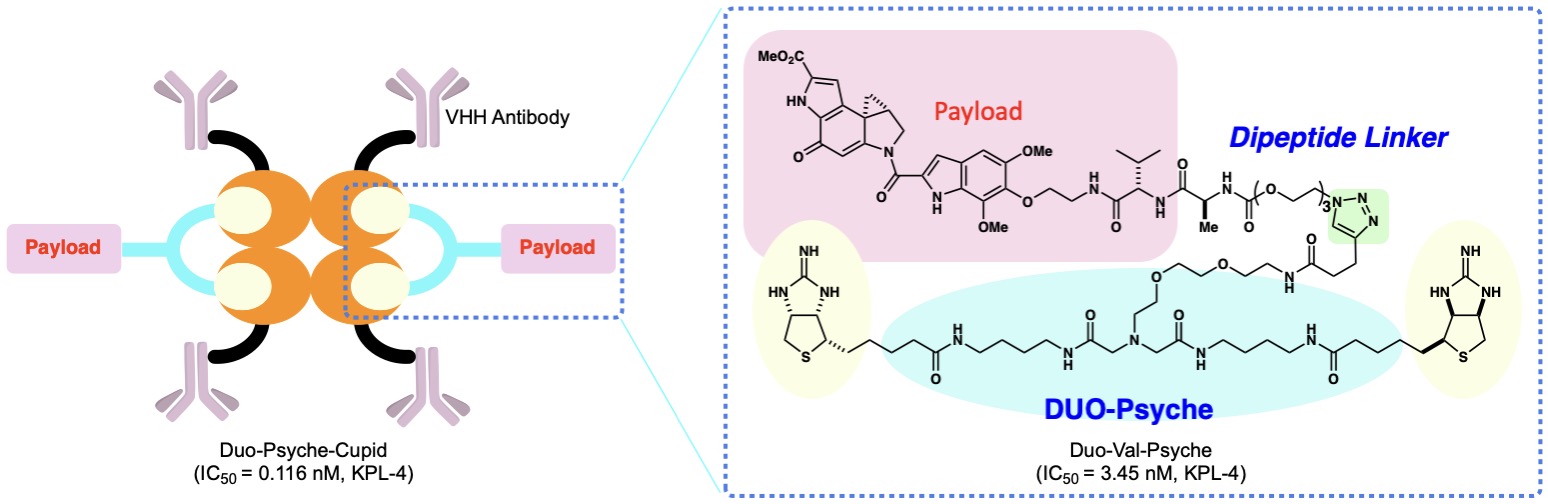
(14) Antibody-mimetic drug conjugate with efficient internalization activity using anti-Her2 VHH and duocarmycin
J. Sakata, T. Tatsumi, A. Sugiyama, A. Shimizu, Y. Inagaki, H. Katoh, T. Yamashita, K. Takahashi,
S. Aki, Y. Kaneko, T. Kawamura, M. Miura, M. Ishii, T. Osawa, T. Tanaka, S. Ishikawa, M. Tsukagoshi,
M. Chansler, T. Kodama, M. Kanai, H. Tokuyama, A. Sugiyama
Protein Expression and Purification, Published online (DOI: 10.1016/j.pep.2023.106375)

(13) Unified Divergent Total Synthesis of Discorhabdin B, H, K, and Aleutianamine via the Late-Stage Oxidative N,S-Acetal Formation
M. Shimomura, K. Ide, J. Sakata, H. Tokuyama
J. Am. Chem. Soc., 145, 18233-18239 (2023) (DOI: 10.1021/jacs.3c06578).

(12) Construction of Tetrahydroquinolines with Spirocyclic Structures at the 4-Position
Y. Wakahara, T. Noro, J. Sakata, H. Ueda, H. Tokuyama
Heterocycles, 105, 438-460 (2022) (DOI:10.3987/COM-22-S(R)16) (Prof. Somsak Ruchirawat's Special Issues).
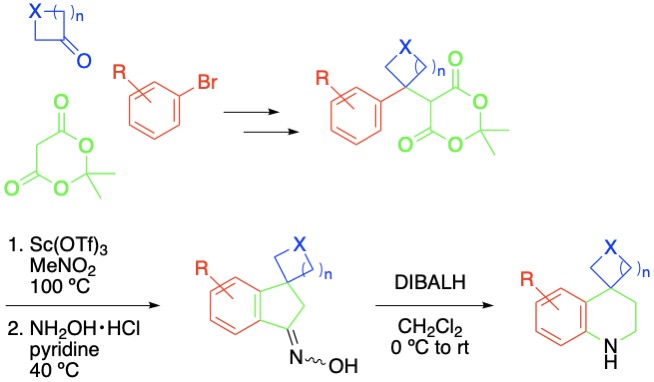
(11) Synthetic Studies on Discorhabdin V: Construction of the A–F Hexacyclic Framework
T. Noro, J. Sakata, H. Tokuyama
Tetrahedron Letters, 81, 153333 (2021) (DOI: 10.1016/j.tetlet.2021.153333).
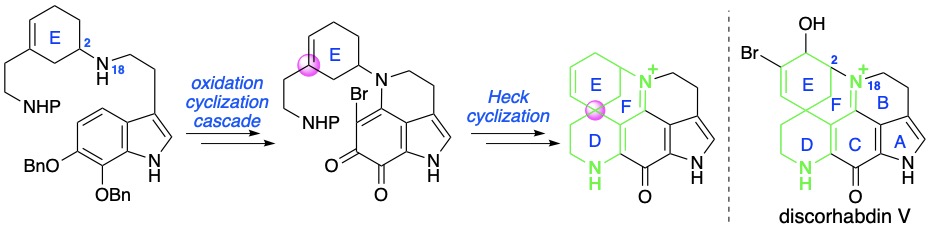
(10) Total syntheses of (-)-emestrin H and (-)-asteroxepin
K. Umeki, Y. Ueda, J. Sakata, H. Tokuyama
Tetrahedron, 76, 131630 (2020) (DOI: 10.1016/j.tet.2020.131630) selected as 2020 editors choice collection
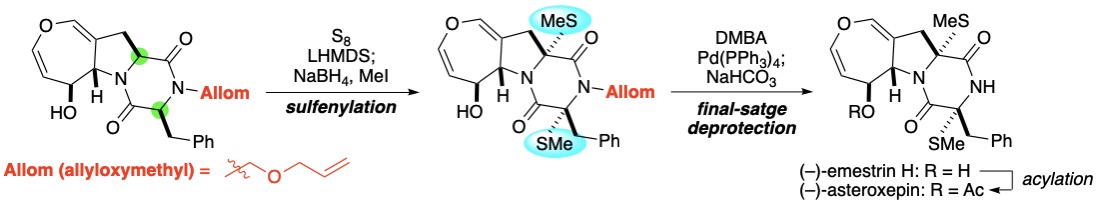
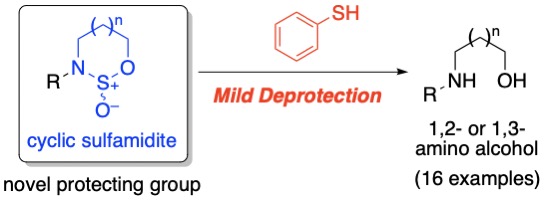
(9) Cyclic Sulfamidite as Simultaneous Protecting Group for Amino Alcohols: Development of a Mild
Deprotection Protocol Using Thiophenol
J. Sakata, K. Akita, M. Sato, M. Shimomura, H. Tokuyama
Chem. Pharm. Bull., 68, 996-1000 (2020) (DOI: 10.1248/cpb.c20-00531).
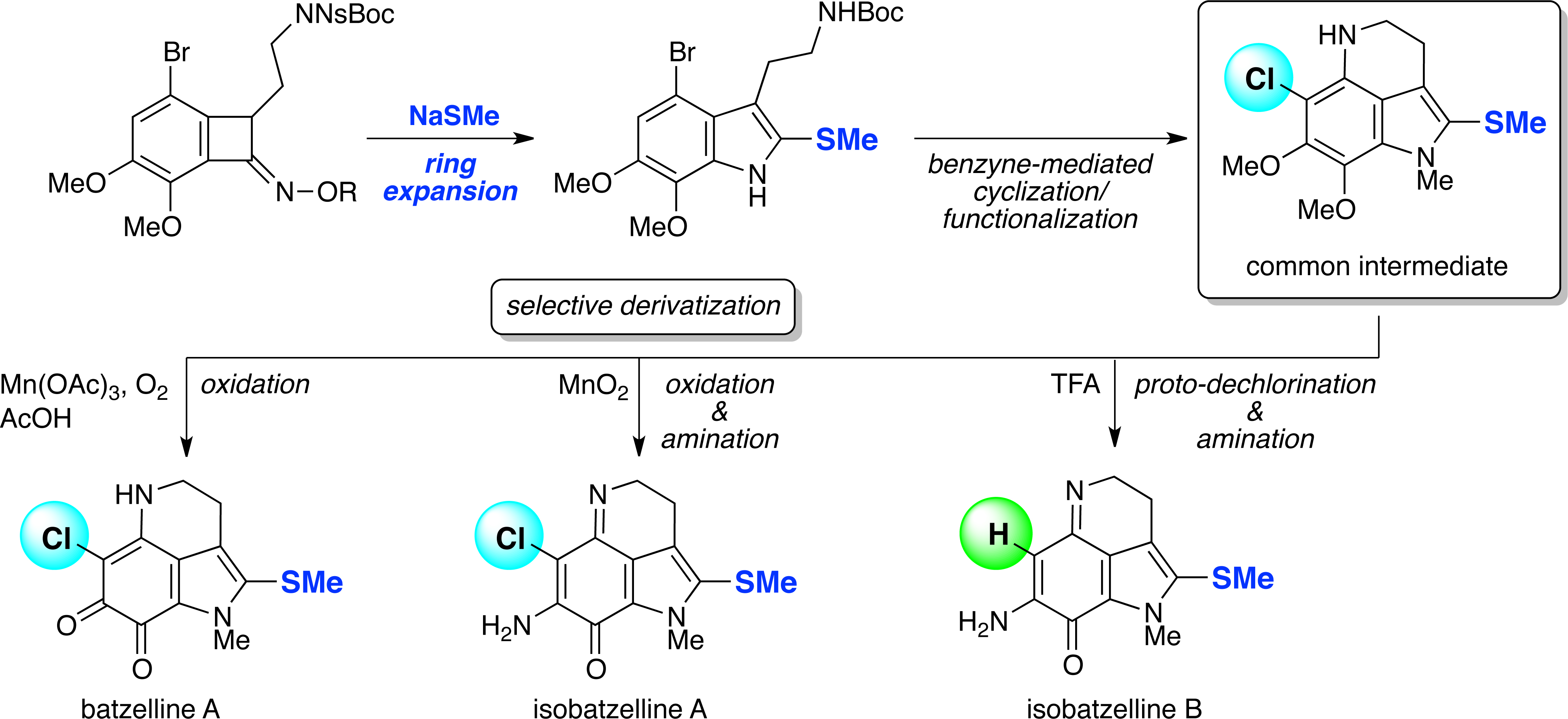
(8) Divergent Total Syntheses of Isobatzellines A/B and Batzelline A
Y. Yamashita, L. Poignant, J. Sakata, H. Tokuyama
Org. Lett., 22, 6239-6243 (2020) (DOI:10.1021/acs.orglett.0c01894). selected as cover picture

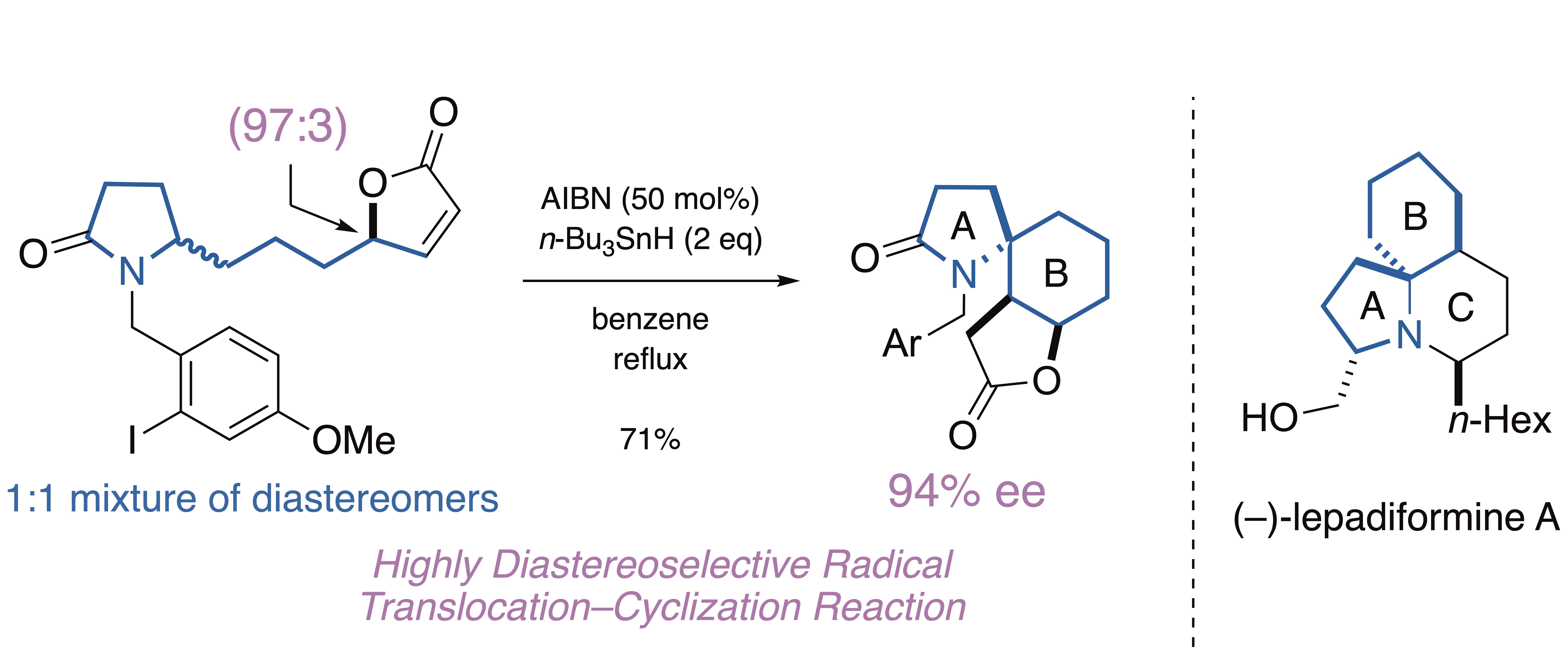
(7) Total Synthesis of (–)-Lepadiformine A via Radical Translocation–Cyclization Reaction
M. Shimomura, M. Sato, H. Azuma, J. Sakata, H. Tokuyama
Org. Lett., 22, 3313-3317 (2020) (DOI:10.1021/acs.orglett.0c00474). selected as cover picture

(6) Fluorescent labeling method re-evaluates the intriguing thermoresponsive behavior of poly(acrylamide-co-acrylonitrile)s (4) Synthetic Studies on Plakinidines (3) One-pot Reductive Allylation of Amides Using a Combination of Titanium Hydride and Allylzinc Reagents: Application (2) Structural determination of (–)-SCH 64874 and hirsutellomycin by semi-synthesis (1) Synthetic Studiy on Naphthospironone A: Construction of Benzobicyclo[3,2,1]octene Skeleton with Oxaspirocycle (1) ひずみエネルギーの解消を駆動力とするアミノ化反応 (1)「Cutting-Edge Organic Synthesis and Chemical Biology of Bioactive Molecules」分担執筆, Yuichi Kobayashi Ed., Springer, 2019, ISBN 978-981-13-6243-9
with upper critical solution temperatures
C. Otsuka, Y. Wakahara, K. Okabe, J. Sakata, M. Okuyama, A. Hayashi, H. Tokuyama, S; Uchiyama
Macromolecules, 52, 7646-7660 (2019) (DOI: 10.1021/acs.macromol.9b00880).
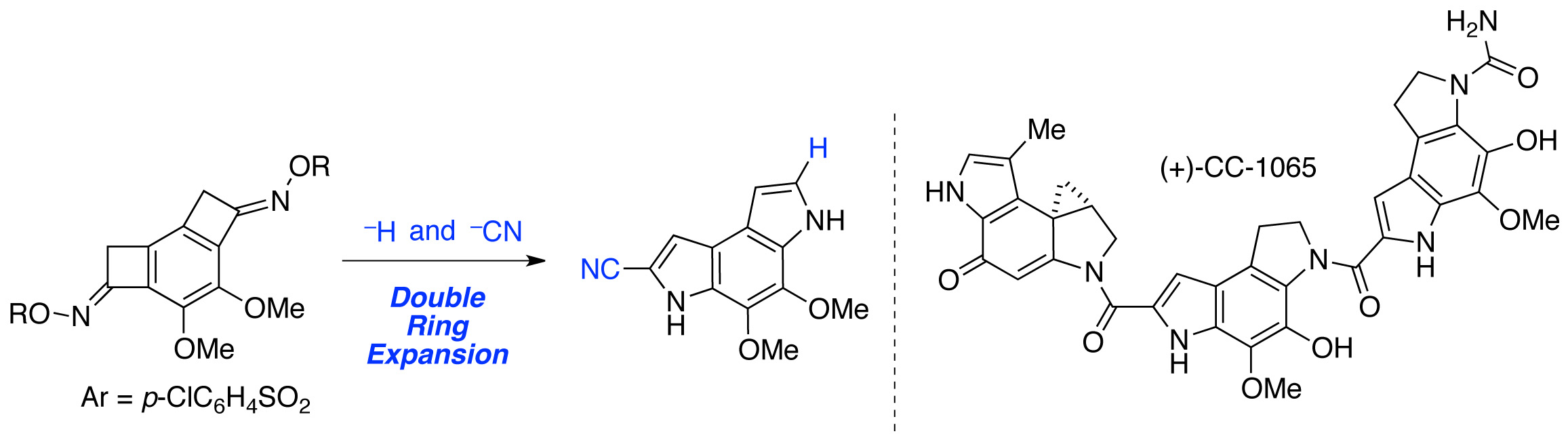
(5) Total Synthesis of (+)-CC-1065 Utilizing Ring Expansion Reaction of Benzocyclobutenone Oxime Sulfonate
T. Imaizumi, Y. Yamashita, Y. Nakazawa, K. Okano, J. Sakata, H. Tokuyama
Org. Lett., 21, 6185-6189 (2019) (DOI: 10.1021/acs.orglett.9b01690). selected as cover picture

T. Satoh, T. Adachi, K. Okano, J. Sakata, H. Tokuyama
Heterocycles, 99, 310-323 (2019) (DOI: 10.3987/COM-18-S(F)26).
to a Total Synthesis of (–)-Castoramine
S. Itabashi, M. Shimomura, M. Sato, H. Azuma, K. Okano, J. Sakata, H. Tokuyama
Synlett, 29, 1786-1790 (2018) (DOI: 10.1055/s-0037-1610435)
H. Tokuyama, K. Yamada, H. Fujiwara, J. Sakata, K. Okano, M. Sappan, M. Isaka
J. Org. Chem., 82, 353-371 (2017)
J. Sakata, Y. Ando, K. Ohmori, K. Suzuki
Org. Lett., 17, 3746-3749 (2015)Review
J.Sakata
Farumashia, 52, 1154 (2016)Book