|
|
-dictizine.PNG) |
Angew. Chem. Int. Ed.,65(4), e21481 (2026) |
|
-15-veratroyl-17-acetyl-19-oxodictizine-2.PNG) |
Angew. Chem. Int. Ed.,65(4), e21481 (2026) |
|
-Aleutianamine.jpeg) |
J. Am. Chem. Soc., 145, 18233-18239 (2023) |
|
-macrocentrine.PNG) |
Angew. Chem. Int. Ed.,65(4), e21481 (2026) |
|
-cochlearenine.PNG) |
Angew. Chem. Int. Ed.,65(4), e21481 (2026) |
|
-Discorhabdin B.jpeg) |
J. Am. Chem. Soc., 145, 18233-18239 (2023) |
|
-Discorhabdin H.jpeg) |
J. Am. Chem. Soc., 145, 18233-18239 (2023) |
|
-Discorhabdin K.jpeg) |
J. Am. Chem. Soc., 145, 18233-18239 (2023) |
|
-Pleiocarpamine.jpeg) |
J. Am. Chem. Soc., 145, 16337-16343 (2023) |
|
-Voacalgine.jpeg) |
J. Am. Chem. Soc., 145, 16337-16343 (2023) |
|
-Bipleiophylline.jpeg) |
J. Am. Chem. Soc., 145, 16337-16343 (2023) |
|
-Dibrevianamide F.jpeg) |
Angew. Chem. Int. Ed., 62(22), e202302404 (2023) |
|
-Naseseazine B.jpeg) |
Angew. Chem. Int. Ed., 62(22), e202302404 (2023) |
|
-Isonaseseazine C.jpeg) |
Angew. Chem. Int. Ed., 62(22), e202302404 (2023) |
|
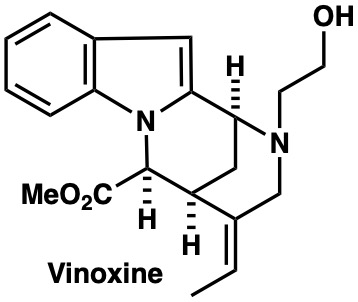 |
Org. Biomol. Chem., accepted for publication (2020), (DOI:10.1039/D2OB00274D) |
|
-15,15'-Bis-epi-eurocristatine.jpeg) |
Angew. Chem. Int. Ed., 62(22), e202302404 (2023) |
|
-Eurocristatine.jpeg) |
Angew. Chem. Int. Ed., 62(22), e202302404 (2023) |
|
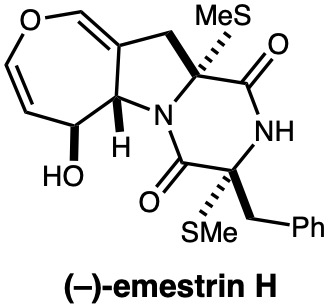 |
Tetrahedron, 76, 131630 (2020) |
|
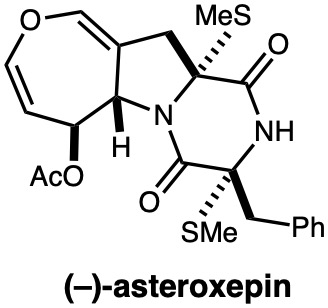 |
Tetrahedron, 76, 131630 (2020) |
|
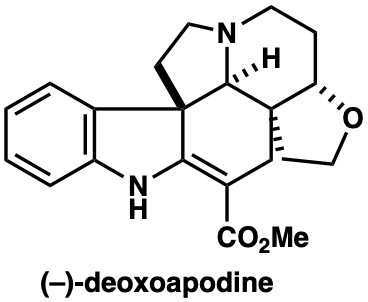 |
Angew. Chem. Int. Ed., 59, 23089 (2020) |
|
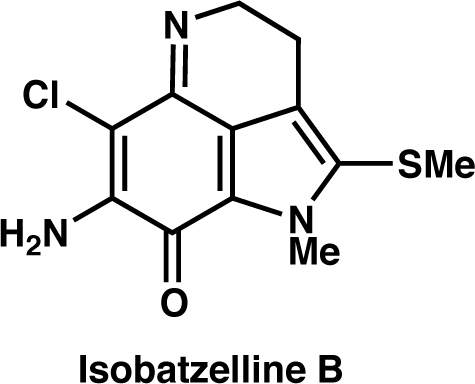 |
Org. Lett., 22, 6239 (2020) |
|
 |
Org. Lett., 22, 6239 (2020) |
|
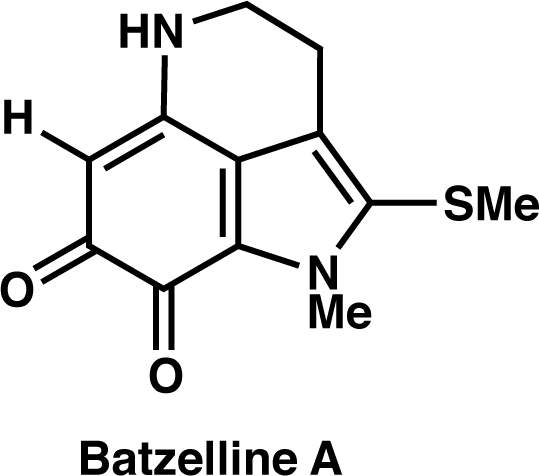 |
Org. Lett., 22, 6239 (2020) |
|
-CC-1065.jpg) |
Org. Lett., 21, 6185-6189 (2019) |
|
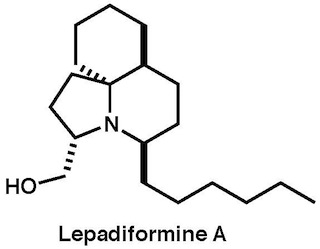
-lepadiformine A.jpg) |
Org. Lett., 22, 3313–3317 (2020) |
|
 |
Chem. Eur. J., accepted article (2020) |
|
-castoramine.jpg) |
Synlett, 29, 1786-1790 (2018) |
|
-pestalazine B.jpg) |
Org. Lett., 21, 4205–4209 (2019) |
|
 |
Org. Lett., 21, 3954–3958 (2019) |
|
-epiamauromine.jpg) |
Org. Lett., 19, 5308–5311 (2017) |
|
-novoamauromine.jpg) |
Org. Lett., 19, 5308–5311 (2017) |
|
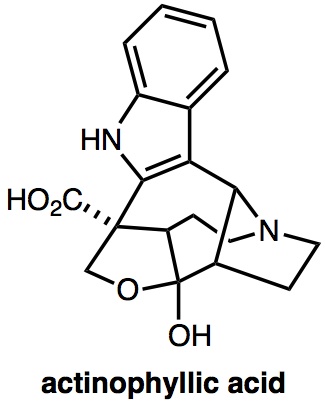 |
Angew. Chem. Int. Ed., 56, 12277-12281 (2017) |
|
-Histrionicotoxin.jpg) |
Angew. Chem. Int. Ed., 56, 1087-1091 (2017) |
|
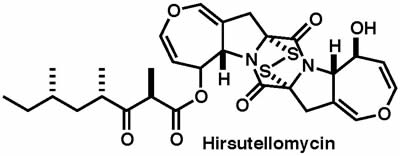 |
J. Org. Chem., 82, 353-371 (2017) |
|
-SCH 64874.jpg) |
J. Org. Chem., 82, 353-371 (2017) |
|
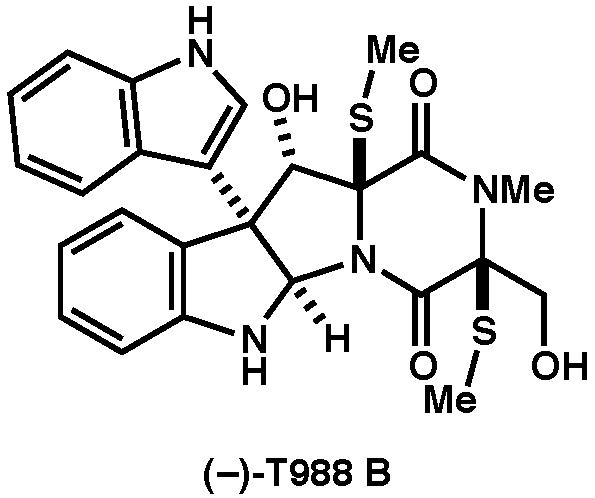 |
Asian J. Org. Chem., 6, 54-58 (2017) |
|
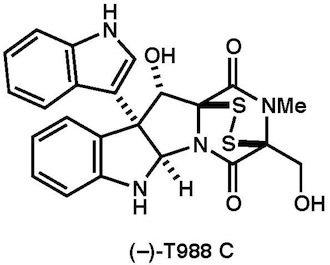 |
Asian J. Org. Chem., 6, 54-58 (2017) |
|
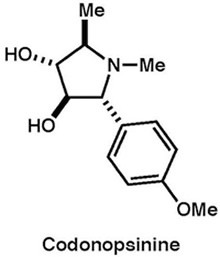 |
J. Antibiot.,69, 253-258 (2016) |
|
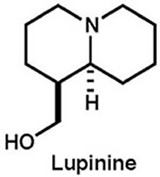 |
Adv. Synth. Catal., 358, 380-385 (2016) |
|
-MPC1001B.jpg) |
Angew. Chem. Int. Ed., 55, 283-287 (2016) |
|
-isoschizogamine .jpg) |
Chem. Eur. J., 21, 16400-16403 (2015) |
|
-Indolactam V.jpg) |
Tetrahedron, 71, 3833-3837 (2015) |
|
-Haouamine B.jpg) |
Angew. Chem. Int. Ed., 53, 13215–13219 (2014) |
|
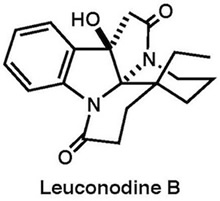 |
Org. Lett., 16, 2526–2529 (2014) Tetrahedron Lett., 79, 131809 (2020) |
|
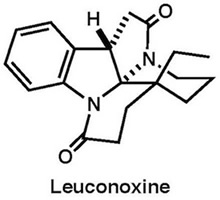 |
Org. Lett., 16, 2526–2529 (2014) Tetrahedron Lett., 79, 131809 (2020) |
|
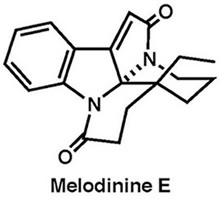 |
Org. Lett., 16, 2526–2529 (2014) |
|
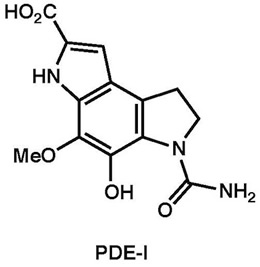 |
Tetrahedron, 69, 10946–10954 (2013)
Chem. Commun., 46, 2641–2643 (2010) |
|
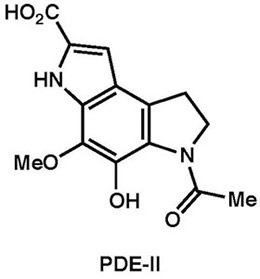 |
Tetrahedron, 69, 10946–10954 (2013)
Chem. Commun., 46, 2641–2643 (2010) |
|
-Rhazinilam.jpg) |
Angew. Chem. Int. Ed., 52, 7168–7171 (2013)
Tetrahedron, 71, 3619-3624 (2015)
Tetrahedron Lett., 79, 131809 (2020) |
|
-Rhazinicine.jpg) |
Angew. Chem. Int. Ed., 52, 7168–7171 (2013) |
|
-Mersicarpine.jpg) |
Chem. Eur. J., 19, 9325–9334 (2013)
Org. Lett., 14, 2320–2322 (2012) |
|
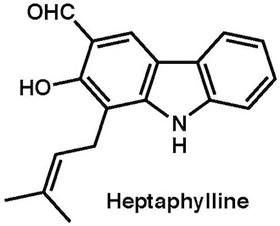 |
Org. Lett., 15, 1946–1949 (2013) |
|
-Aspidophytine.jpg) |
Tetrahedron, 69, 89–95 (2013) |
|
-Cimicidine.jpg) |
Tetrahedron, 69, 89–95 (2013)
|
|
-Cimicine.jpg) |
Tetrahedron, 69, 89–95 (2013) |
|
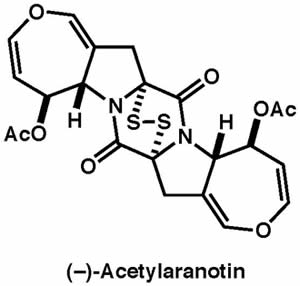 |
Angew. Chem. Int. Ed., 51, 13062–13065 (2012) |
|
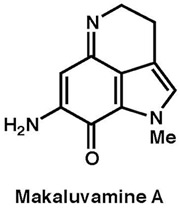 |
Tetrahedron, 68, 9376–9383 (2012) |
|
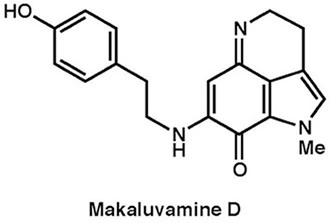 |
Tetrahedron, 68, 9376–9383 (2012) |
|
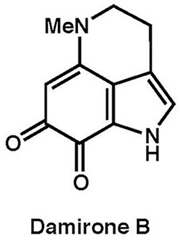 |
Tetrahedron, 68, 9376–9383 (2012) |
|
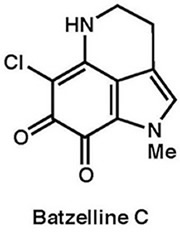 |
Tetrahedron, 68, 9376–9383 (2012)
RSC Adv., 2, 5147-5149 (2012) |
|
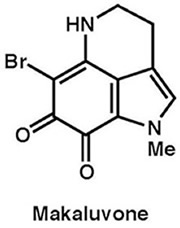 |
Tetrahedron, 68, 9376–9383 (2012) |
|
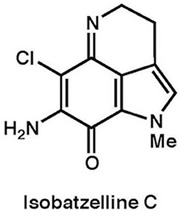 |
Tetrahedron, 68, 9376–9383 (2012)
RSC Adv., 2, 5147-5149 (2012) |
|
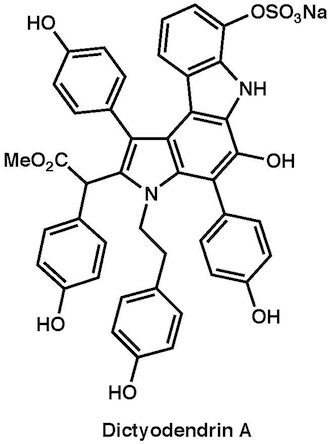 |
Chem. Asian J., 6, 560–572 (2011)
Angew. Chem. Int. Ed., 49, 5925–5929 (2010) |
|
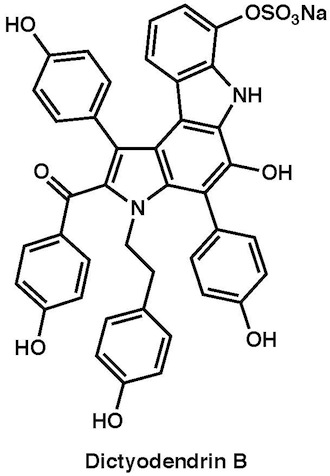 |
Chem. Asian J., 6, 560–572 (2011)
Angew. Chem. Int. Ed., 49, 5925–5929 (2010) |
|
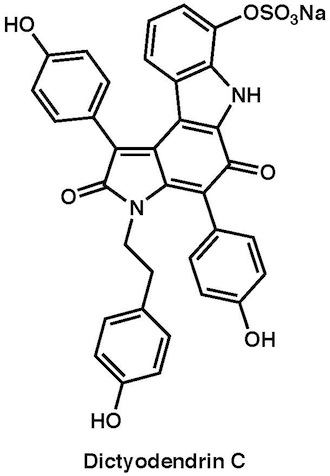 |
Chem. Asian J., 6, 560–572 (2011) |
|
 |
Chem. Asian J., 6, 560–572 (2011) |
|
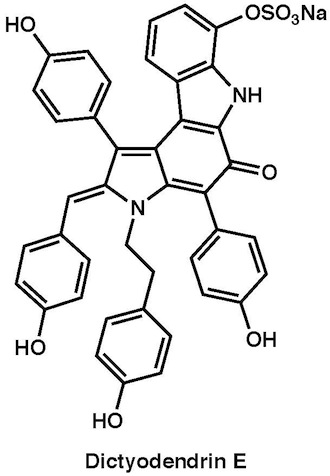 |
Chem. Asian J., 6, 560–572 (2011) |
|
-Petrosin.jpg) |
Org. Lett., 12, 5196–5199 (2010) |
|
 |
Synlett, 822–826 (2010) |
|
-Haplophytine.jpg) |
Angew. Chem. Int. Ed.,55, 15157–15161 (2016)
Angew. Chem. Int. Ed., 48, 7600–7603 (2009) |
|
|


-Aleutianamine.jpeg)
-Discorhabdin B.jpeg)
-Discorhabdin H.jpeg)
-Discorhabdin K.jpeg)
-Pleiocarpamine.jpeg)
-Voacalgine.jpeg)
-Bipleiophylline.jpeg)
-Dibrevianamide F.jpeg)
-Naseseazine B.jpeg)
-Isonaseseazine C.jpeg)

-15,15'-Bis-epi-eurocristatine.jpeg)
-Eurocristatine.jpeg)






-CC-1065.jpg)
-lepadiformine A.jpg)

-castoramine.jpg)
-pestalazine B.jpg)

-epiamauromine.jpg)
-novoamauromine.jpg)

-Histrionicotoxin.jpg)

-SCH 64874.jpg)




-MPC1001B.jpg)
-Indolactam V.jpg)
-Haouamine B.jpg)





-Rhazinilam.jpg)
-Rhazinicine.jpg)
-Mersicarpine.jpg)

-Aspidophytine.jpg)
-Cimicidine.jpg)
-Cimicine.jpg)












-Petrosin.jpg)

-Haplophytine.jpg)