生合成中間体の多能性を活用する
Teigo Asai1,*, Kento Tsukada1, Satomi Ise1, Naoki Shirata1, Makoto Hashimoto2, Isao Fujii2 Katsuya Gomi3, Kosuke Nakagawara4, Eiichi N. Kodama4 and Yoshiteru Oshima1,*
1Graduate School of Pharmaceutical Sciences, Tohoku University, Aoba-yama, Aoba-ku, Sendai, Japan. 2School of Pharmacy, Iwate Medical University, Nishitokuta, Yahaba, Iwate, Japan. 3Graduate School of Agricultural Science, Tohoku University, Sendai, Japan. 4Graduate School of Medicine/Tohoku Medical Megabank Organization, Tohoku University, Sendai, Japan.
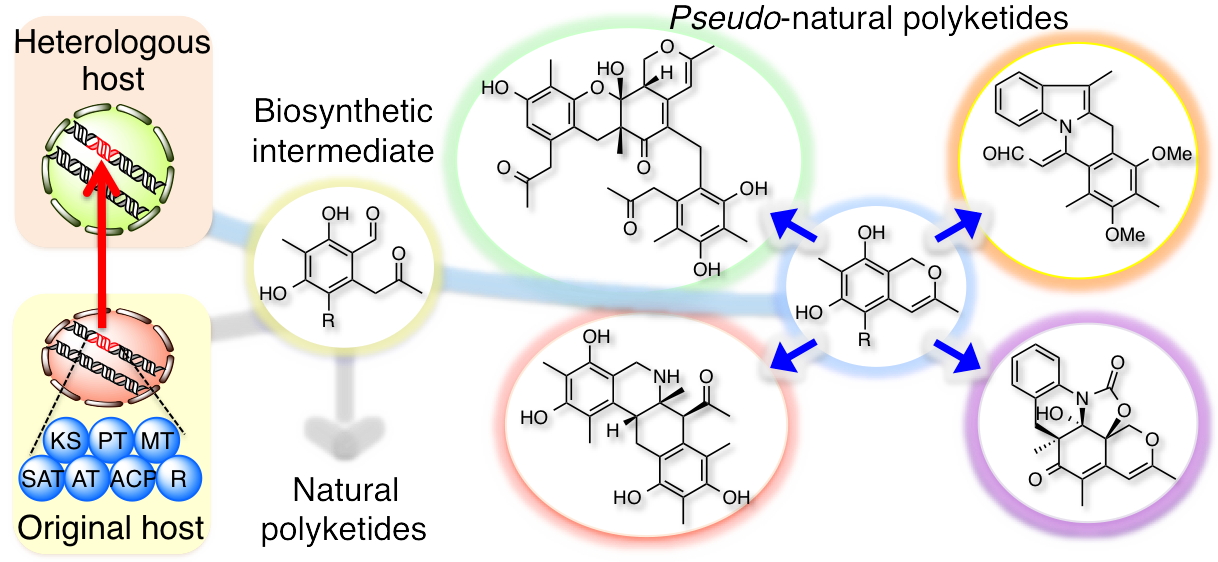
The structural complexity and diversity of natural products make them attractive sources for potential drug discovery, with their characteristics being derived from the multi-step combination of enzymatic and non-enzymatic conversions of intermediates in each biosynthetic pathway. Intermediates that exhibit multipotent behaviour have great potential for use as starting points in diversity-oriented synthesis. Inspired by the biosynthetic pathways that form complex metabolites from simple intermediates, we developed a semi-synthetic process that combines heterologous biosynthesis and artificial diversification. The heterologous biosynthesis of fungal polyketide intermediates led to the isolation of novel oligomers and provided evidence for ortho-quinonemethide equivalency in their isochromene form. The intrinsic reactivity of the isochromene polyketide enabled us to access various new chemical entities by modifying and remodelling the polyketide core and through coupling with indole molecules. We thus succeeded in generating exceptionally diverse pseudo-natural polyketides through this process and demonstrated an advanced method of using biosynthetic intermediates.